In a landmark judgment with far-reaching implications for the dairy and livestock ecosystem, the Delhi High Court has set aside key directives issued by the Food Safety and Standards Authority of India that sought to regulate animal feed. The Court held that under the Food Safety and Standards Act, 2006, FSSAI’s jurisdiction is limited strictly to food meant for human consumption and does not extend to cattle or animal feed. This effectively invalidates multiple advisories issued between 2019 and 2021, including restrictions on the use of certain ingredients such as meat and bone meal (MBM) in feed for milk- and meat-producing animals.
The ruling brings much-needed clarity to a long-debated regulatory overlap, particularly where FSSAI had also attempted to mandate compliance with Bureau of Indian Standards norms for animal feed—an area the Court deemed beyond its statutory authority. While the decision is being welcomed by segments of the feed industry for reducing compliance ambiguity, it simultaneously opens up a regulatory gap regarding quality control, safety standards, and traceability of feed inputs that directly influence milk quality and food safety outcomes.
From a dairy sector perspective, this development could reshape feed formulation strategies, especially around protein sourcing, while shifting the onus of regulation more decisively toward the Department of Animal Husbandry, BIS, and state-level authorities. The broader implication remains critical: as India scales its dairy value chain and moves toward higher-value products, ensuring robust and clearly defined governance of animal nutrition will be essential.
Core Findings of the High Court Ruling
-
-
Statutory Limitation: The Bench (Chief Justice Devendra Kumar Upadhyaya and Justice Tejas Karia) held that the Food Safety and Standards Act, 2006 is strictly anchored to food intended for human consumption. Since the Act specifically excludes animal feed from its definition of "food," the FSSAI cannot extend its reach to the livestock feed sector.
-
Quashed Regulation (Note c): The court struck down Note (c) of Regulation 2.5.2 of the FSS (Food Products Standards and Food Additives) Regulations, 2011. This note had prohibited using meat or bone meal (of bovine or porcine origin) in feed for milk-producing animals.
-
BIS Certification Not Mandatory: The court invalidated FSSAI directives (from 2019, 2020, and 2021) that made BIS (Bureau of Indian Standards) certification mandatory for commercial feeds. The court clarified that BIS standards are generally voluntary and can only be made mandatory by the Central Government under the BIS Act, not by an FSSAI order.
-
Ultra Vires: The court declared the FSSAI’s attempt to regulate feed as ultra vires (beyond its legal power), stating that if the regulator wanted to control what animals eat to ensure human food safety, it would require a Parliamentary amendment to the parent Act.
-
Who Will Guard Milk Safety Now?
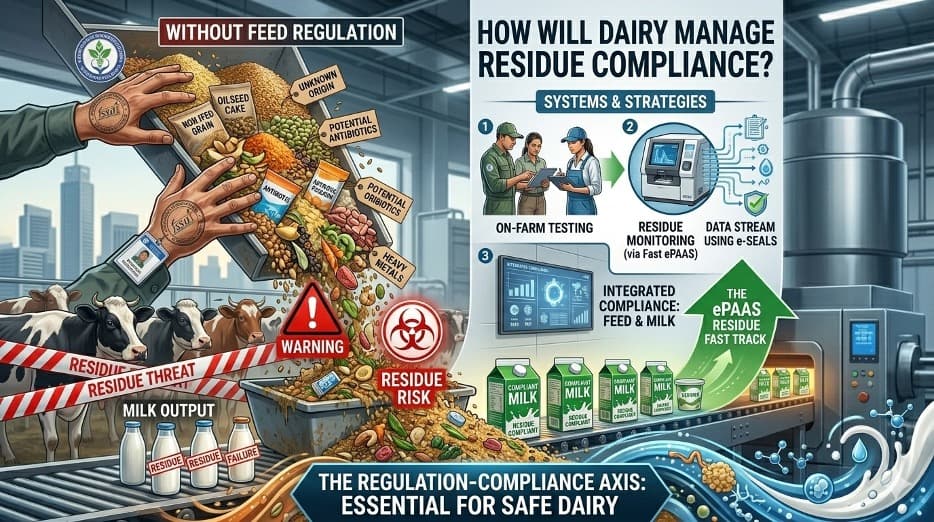
The recent ruling by the Delhi High Court, as discussed above, setting aside the Food Safety and Standards Authority of India directives on animal feed, has brought regulatory clarity—but also a serious and somewhat uncomfortable question for India’s dairy sector: who will now take responsibility for controlling contaminants that originate in feed but end up in milk?
This concern is not theoretical. It is rooted in hard evidence from the National Milk Adulteration Survey 2018, where aflatoxin contamination emerged as one of the most critical food safety risks, detected in about 5.7% of milk samples. Aflatoxin M1 in milk is directly linked to Aflatoxin B1 in cattle feed—primarily from contaminated maize, oil cakes, and improperly fermented silage. The dairy value chain, therefore, is only as strong as its weakest upstream input: feed.
It was in this context that FSSAI had attempted to step in—nudging the ecosystem toward stricter compliance, including alignment with Bureau of Indian Standards norms for compounded feed. While the legal position may now be settled—that FSSAI’s mandate under the Food Safety and Standards Act, 2006 does not extend to animal feed—the food safety implications remain unresolved.
Milk is not an isolated product; it is a biological output of what the animal consumes. When contaminated feed enters the system, the risk seamlessly transfers into milk, and from there into the human food chain. In a country that prides itself on being the world’s largest milk producer, even a 5–6% contamination level is not a marginal issue—it is a systemic vulnerability.
The current judgment risks creating a regulatory vacuum at precisely the point where vigilance is most needed. Feed manufacturers may find compliance easier, but the absence of a clearly empowered authority to enforce standards on aflatoxin levels in feed ingredients, compounded feed, and emerging formats like commercial silage could weaken the safety net. The challenge is even more acute given the increasing use of maize silage and total mixed rations (TMR) in organized dairy operations, where mycotoxin risks, if not managed scientifically, can scale rapidly.
At a broader level, this also introduces a subtle policy contradiction. India has consistently maintained a cautious stance on dairy imports, particularly from countries like the United States, where regulatory frameworks such as those under the U.S. Food and Drug Administration permit the use of certain animal-derived feed ingredients like fish meal and meat and bone meal. These concerns have often been cited in safeguarding domestic dairy standards. Yet, at the same time, the dilution of regulatory oversight on similar inputs within our own feed ecosystem raises an important question of alignment—between what we seek to prevent at our borders and what we are willing to monitor within.
This is not to argue against the Court’s interpretation of the law—it is to highlight the urgent need for regulatory realignment. If FSSAI cannot regulate feed, then the responsibility must be explicitly and effectively anchored with agencies such as the Department of Animal Husbandry, in coordination with BIS and state regulators. More importantly, there must be a seamless linkage between feed standards and food safety outcomes, because the two are inseparable in dairy.
The way forward calls for three immediate actions. First, a national framework for mycotoxin control in feed, with enforceable limits and surveillance. Second, mandatory testing protocols for high-risk feed ingredients and silage. Third, a traceability mechanism that links feed quality to milk procurement systems, especially in organized dairies.
India has made remarkable progress in building a sustainable and inclusive dairy ecosystem. But as we move toward higher value addition and global competitiveness, food safety cannot be treated in silos. The question before policymakers is simple yet critical: if not FSSAI, then who ensures that what the animal eats does not compromise what the consumer drinks?
Source : Blog by Kuldeep Sharma Chief editor Dairynews7x7 April 9th 2026
News source : Livelaw. Lawtrend
Summary of Quashed Directives
The ruling specifically nullified the following FSSAI orders:
-
Directive dated 10.12.2019
-
Directive dated 27.01.2020
-
Directive dated 01.01.2021