The draft amendment issued by Food Safety and Standards Authority of India under the Food Safety and Standards (Contaminants, Toxins and Residues) Regulations, 2024 is far more than a routine updation of pesticide residue limits. It is a strong signal that the future competitiveness of Indian dairy will increasingly depend not only on milk production and processing capacities, but on residue assurance, feed traceability and globally benchmarked food safety systems.
From cottonseed cake and maize silage to soybean meal, paddy straw and green fodder, the regulation brings the entire “feed-to-milk” ecosystem under sharper scrutiny. With several pesticide MRLs revised for milk, milk products and milk fat, the industry now faces the challenge — and opportunity — of transitioning toward a more integrated residue-risk management approach aligned with global expectations.
As premium export markets become increasingly sensitive to pesticide residues, traceability and sustainability-linked compliance, this draft notification could eventually become a turning point for India’s dairy exports, particularly in high-value ingredients, infant nutrition and premium dairy categories. At the same time, implementation challenges remain significant in India’s smallholder-driven dairy ecosystem where feed sourcing is fragmented and awareness regarding pesticide withdrawal practices remains limited.
The industry therefore needs to proactively engage on this notification before it is finalized and gazetted. Dairy processors, cooperatives, feed manufacturers, farmer organizations, exporters, laboratories and technical experts should carefully examine the proposed MRLs, evaluate practical implementation challenges, assess alignment with international standards and submit their recommendations, scientific inputs and operational concerns to Food Safety and Standards Authority of India within the consultation period. Constructive industry participation at this stage will be critical to ensure that the final regulation remains both scientifically robust and practically implementable for India’s unique dairy ecosystem.
One of the most important provisions in the draft regulation is the proposal that any pesticide not registered under the Insecticides Act, 1968 shall attract a default tolerance limit of 0.01 mg/kg, except in spices and culinary herbs where the tolerance limit shall be 0.1 mg/kg. This single provision has major implications for dairy because residues in milk do not generally originate from direct application on milk itself, but through the feed and fodder ecosystem surrounding dairy animals. Cottonseed cake, maize, soybean meal, paddy straw, green fodder, sugarcane by-products and even contaminated water become the indirect pathways through which pesticide residues finally reach milk and milk products.The notification includes revised or newly prescribed MRLs for a large number of pesticides that are directly or indirectly relevant to the dairy value chain. Among the important molecules impacting milk and milk products are Acephate with a proposed limit of 0.02 mg/kg, Acetamiprid at 0.2 mg/kg, Azoxystrobin at 0.01 mg/kg, Carbendazim at 0.1 mg/kg, Carbofuran at 0.05 mg/kg, Chlorpyrifos at 0.02 mg/kg, Cyhalothrin at 0.2 mg/kg, Cypermethrin at 0.05 mg/kg, Deltamethrin at 0.05 mg/kg, Dimethoate at 0.05 mg/kg, Ethion at 0.5 mg/kg, Fipronil at 0.02 mg/kg, Flubendiamide at 0.1 mg/kg and Dinotefuran at 0.1 mg/kg in milk and milk products. Many of these pesticides are extensively linked with crop protection practices in feed crops, storage pest management, vector control in dairy farms and ectoparasite management in livestock.
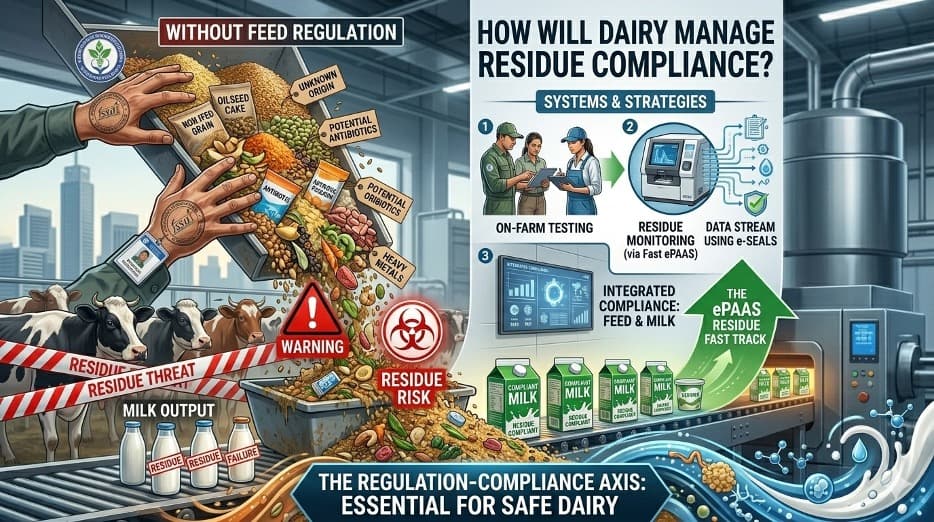
Feed to Milk Surviellance Approach
The biggest takeaway for the dairy industry is that food safety management can no longer remain confined to milk testing at the dairy plant gate. Traditionally, most dairy processors focused primarily on adulteration, antibiotics and aflatoxin M1 surveillance. However, this notification pushes the sector toward an integrated “feed-to-milk” surveillance approach. In the coming years, feed traceability may become as important as milk traceability itself. Dairies will increasingly have to monitor pesticide practices in the cultivation of cotton, maize, soybean, paddy and fodder crops because the residue burden entering the animal feed chain may eventually determine the residue profile of milk.
This notification also comes at a strategically important time when India is attempting to position itself more aggressively in global dairy trade. International dairy markets, particularly premium markets in the European Union, Japan, United States, Australia and New Zealand, are increasingly residue-sensitive. Importing countries are now demanding not only compliance with Codex standards but also sophisticated multi-residue screening protocols, traceability systems and sustainability-linked food safety assurances. Until now, Indian dairy exports have largely catered to SMP-deficit regions, neighbouring countries, institutional buyers and price-sensitive markets where residue scrutiny was relatively limited. But future export growth, especially in high-value dairy ingredients, infant nutrition, functional dairy products and premium consumer dairy categories, will require residue assurance systems comparable to global benchmarks.
Another critical aspect often overlooked is that many pesticide molecules are lipophilic in nature, meaning they tend to accumulate in fat. This makes products such as ghee, butter, cream, khoa, cheese and high-fat milk powders more vulnerable from a residue perspective. The draft notification separately specifies residue limits for milk, milk products and milk fat in several cases, clearly indicating regulatory recognition of this concentration effect. For export-oriented Indian dairies, this becomes especially important because international residue testing frequently targets fat-rich dairy matrices.
From a regulatory science perspective, the draft regulation indicates that India is steadily moving toward a more risk-based food safety framework aligned with international trends under Codex Alimentarius and SPS-driven trade systems. The emphasis is gradually shifting from reactive end-product testing toward preventive residue-risk management across the entire food chain. This is a positive development for the long-term credibility of Indian food exports, including dairy.
Farmer awareness will emerge as one of the most critical pillars of future compliance. Farmers need practical training regarding approved pesticide molecules, withdrawal periods, fodder contamination risks and safe feed storage practices. At present, the understanding of how pesticide residues move from field to feed and eventually into milk remains extremely poor at ground level. Unless this knowledge gap is addressed, enforcement alone may create disruption without delivering meaningful improvement in residue management.
The notification also highlights the urgent need for stronger integration between feed regulation and dairy food safety regulation in India. Milk safety can no longer be viewed independently of feed safety. Greater convergence will be required between Food Safety and Standards Authority of India, animal husbandry departments, feed regulators and pesticide authorities to create a practical and science-based implementation ecosystem.
In reality, this draft notification is not merely about revising pesticide residue limits. It is an indication that the future competitiveness of Indian dairy may increasingly depend not only on milk production volumes or processing capacity, but on residue assurance, traceability and clean-label credibility. The dairy processors who start investing today in feed surveillance, farmer education and residue-risk management are likely to emerge tomorrow as the most credible suppliers for both premium domestic consumers and global dairy markets.
Source : FSSAI draft notification review by Kuldeep Sharma Chief editor Dairynews7x7
Download the draft notification here
#DairyIndustry #FeedSafety #FSSAI #MilkSafety #ResidueManagement #FeedTraceability #PesticideResidues #IndianDairy #FoodSafety #RegulatoryAffairs #DairyExports #MilkQuality #FeedRegulation #Codex #CleanMilk